|
9/26/2023 0 Comments No molecular orbital diagram 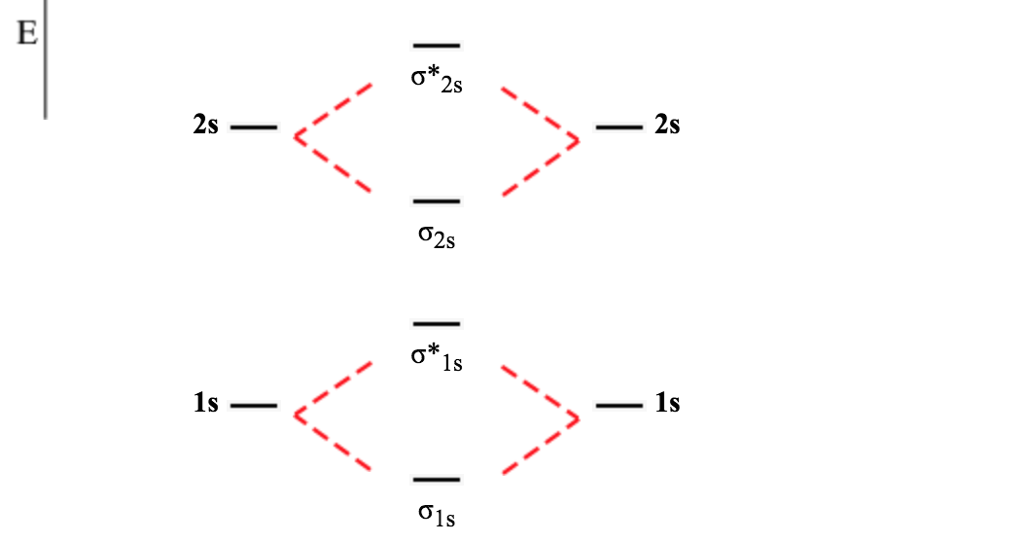
A MO diagram dispenses with the need for resonance. A molecular orbital diagram describes the bonding in terms of constructive and destructive overlap of atomic orbitals. Item 1: Part B Based on the molecular orbital diagram for NO. To answer this question, we nned molecular orbital theory. Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin. By drawing molecular orbital diagrams for, ,, , and, predict which of these homonuclear. The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). Molecular Orbital Diagrams, Bond Order, and Number of Unpaired Electrons Solution We draw a molecular orbital energy diagram similar to that shown in link. Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. Find predesigned No Molecular Orbital Diagram Ppt Powerpoint Presentation Layouts Aids Cpb PowerPoint templates slides, graphics, and image designs provided. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. (c) Which of the molecular orbitals in BC do not have a planar node along the internuclear axis (d) Which has the stronger BC bond, BC or BC. For chem videos, quizzes and more download Chemistry X for free on the App Store Correlation Diagrams - by considering the positions and energies of electr. (b) Write the molecular orbital configuration for the valence electrons in BC and in BC. Molecular orbital (MO) diagram for N2 and N2- 2 Do chemists use molecular orbital theory to explain physical characteristics of a compound (i.e. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. (a) Draw a MO diagram for the valence electrons of BC. Needs multiple structures to describe resonance Predicts the arrangement of electrons in molecules Predicts molecular shape based on the number of regions of electron density \): Comparison of Bonding Theories Valence Bond TheoryĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
